|
 |
- Search
| Korean J Neuromuscul Disord > Volume 15(2); 2023 > Article |
|
ABSTRACT
Background
The younger generation in Korea easily exposed to nitrous oxide (N2O) under the name “Happy Balloon” may abuse it. N2O can irreversibly oxidize vitamin B12 and cause abnormal hematopoiesis or nervous system toxicity such as subacute combined degeneration (SCD). The objective of this study was to assist in early diagnosis of N2O-induced SCD of spinal cord by characterizing its clinical manifestations.
Methods
Four patients with myelopathy after abusing N2O were enrolled. To characterize N2O-induced myelopathy, previously reported cases of N2O-induced SCD were searched through PubMed and KoreaMed. Collected cases and our four patients were analyzed.
Results
A total of 30 patients with N2O-induced myelopathy (26 males and 4 females with mean age of 24 years) were analyzed. These patients took a median dose of 650 canisters for a median duration of 3.5 months. All patients presented sensory disturbances, which involved the lower extremities more frequently (100%) than the upper extremities (63.3%). Gait ataxia (76.7%), weakness in the upper (23.3%) and lower (36.7%) extremities, bladder symptoms (26.7%), Rhomberg sign (43.3%), and Lhermitte’s phenomenon (10.0%) were observed. Serum vitamin B12 levels were decreased in many (63.3%) patients and homocysteine levels were elevated in all. Of 20 patients who underwent magnetic resonance imaging (MRI) of the spine, 19 had abnormal findings. Three patients presented with contrast enhancement in lesions.
Conclusions
We strongly recommend that history of N2O abuse should be asked for young patients with suspected myelopathy, especially those presenting with gait ataxia and sensory disturbances suggesting posterior column dysfunction and those presenting long-segment lesion involving the upper cervical cord on MRI.
Nitrous oxide is one of the most widely used anesthetic agents worldwide owing to its potent analgesic effects. It is available in the form of whipped cream canisters [1]. Recently, under the name “"Happy Balloon,” N2O is used as a recreational drug in Korea owing to its euphoric qualities. Therefore, the younger generation easily exposed to it may abuse it. Nitrous oxide converts vitamin B12 to its inactive form by irreversible oxidation, causing clinical features of vitamin B12 deficiency [1]. Vitamin B12 is required for DNA synthesis and methylation. This process is important for the synthesis and maintenance of myelin sheaths. Its deficiency can lead to pathological conditions such as subacute combined degeneration (SCD), including myelopathy, neuropathy, neuropsychiatric disturbance, and abnormal hematopoiesis [2,3]. Symptoms commonly present include paresthesia, numbness, abnormal proprioception, and vibratory sensation. They might also include motor weakness, clumsiness, and gait impairment [4,5]. Therefore, the objective of this study was to collect and summarize reported cases, including our cases, to characterize clinical features, laboratory results, and imaging findings of N2O-induced SCD of spinal cord for early detection.
We conducted a search of PubMed and KoreaMed databases to identify studies on N2O myelopathy with keywords of nitrous oxide or laughing gas or N2O myelopathy or subacute degeneration of the spinal cord or SCD or myeloneuropathy or spinal cord. Studies were limited to human and case reports or case series published before June 2022. Thirty cases of N2O-induced myelopathy, including our four cases, were collected for the analysis. Our four patients were recruited from January 2018 to June 2022 at one university hospital. This retrospective study was approved by the local Human Research Protection Office/Institutional Review.
We evaluated baseline characteristics (including age, sex, exposure duration, and dose of N2O before symptom onset) and clinical features (including laboratory, electrophysiology and imaging tests, treatment, and recovery time). Recovery time was defined as the duration between discontinuation of N2O inhalation and improvement to a normal neurological status. Laboratory tests (including serum vitamin B12 and homocysteine levels) and magnetic resonance imaging (MRI) of the spine were performed. Data were analyzed using SPSS software version 25.0 (IBM Co., Armonk, NY, USA). Results are expressed as mean with standard error or median with range. Statistical significance was set at p < 0.05.
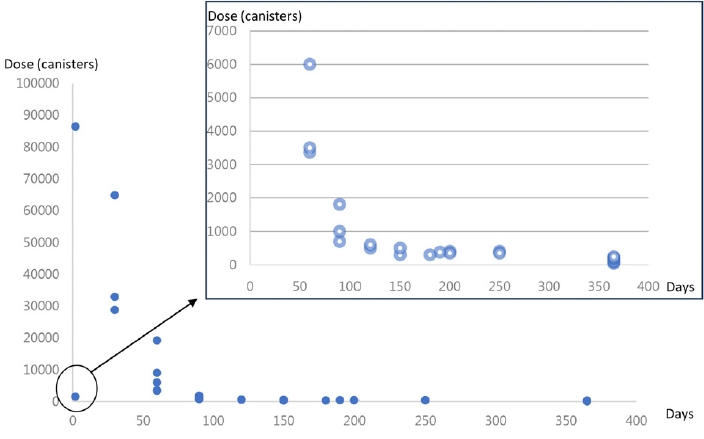
Clinical characteristics of the 30 patients are summarized in Table 1. The average age of patients was 24 years (range, 17-36 years). Myelopathy was induced by a median dose of 650 canisters (range, 40-86,400 canisters) for a median duration of 3.5 months (range, 1-36 months) (Fig. 1). Sensory disturbance was the most frequent presentation, occurring more often in the lower extremities (100%, n = 30) than in the upper extremities (63.3%, n = 19). Gait ataxia (76.7%, n = 23) was the second most common type of ataxia. Weakness in the upper (23.3%, n = 7) and lower (36.7%, n = 11) extremities, bladder symptoms (26.7%, n = 8), Rhomberg sign (43.3%, n = 13), and Lhermitte’s phenomenon (10.0%, n = 3) were also observed.
Serum vitamin B12 levels decreased in only 65.5% (n = 19) of patients, indicating that subacute degeneration of the spinal cord in this situation was triggered by functional rather than absolute vitamin B12 deficiency (Table 2). Homocysteine levels were elevated in all serum samples assayed. Most (29/30, 96.7%) patients recovered functional activity perfectly after several doses of vitamin B12 supplementation (cobalamin 1,000 mcg IM/SC daily for 5 days followed by 1,000 mcg/week for 5 weeks) within several months (median: 4.9 months; interquartile range: 2.0-14.4 months) [6]. Homocysteine level was also normalized (mean 13.2 ± 4.3 mcmol/L).
Among 30 cases, electrophysiology test was performed for 9 cases, including our four patients. All abnormal findings were confirmed in posterior tibial sensory evoked potential. Five patients had an abnormality in median nerve sensory evoked potential and one patient had sensorimotor polyneuropathy in nerve conduction study (Table 2).
The significance of this study is that it broadens the clinical perspective of serious but treatable myelopathy by N2O abuse that is easily overlooked clinically. In this study, the majority (86.66%) of patients with N2O-induced myelopathy were young men. The duration of exposure to N2O was from several weeks to several years. While the relationship between myelopathy and the dose or duration of N2O inhalation remains unclear [7], this study reveals that more than 40 canisters (median dose of 650) can induce myelopathy.
This study found that the most common clinical symptom was sensory impairment in the lower extremities and that serum homocysteine converted by active vitamin B12 was more sensitive than serum vitamin B12 levels. This indicates that subacute degeneration of the spinal cord is caused by functional deficiency rather than absolute vitamin B12 deficiency [8].
Nitrous oxide is widely used as an anesthetic gas to make whipped cream and coffee for recreational purposes [9,10]. Currently, N2O or laughter gas is readily available and increasingly abused. Inhalation of N2O can result in almost immediate psychotropic effects, including euphoria, giggling, sound distortion, and mild hallucinations [6].
Nitrous oxide can convert vitamin B12 to its inactive form by irreversible oxidation, causing clinical features of vitamin B12 deficiency. This irreversible reaction subsequently causes inhibition of methionine synthase enzyme and L-methyl-malonyl-coenzyme A mutase because vitamin B12 is a cofactor for these two enzymes. This process prevents the conversion of homocysteine to methionine and methylmalonic acid to succinyl-CoA, resulting in increased concentrations of homocysteine and methylmalonic acid. Methionine, a precursor of S-adenosylmethionine, is critical for myelin production. Methylmalonic acid can contribute to increased destabilization of the myelin sheath [7]. Therefore, reduced methionine levels and increased methylmalonic acid levels may cause demyelinating myeloneuropathy accompanied by axonal degeneration in central and peripheral nervous systems [4,11].
Long-segment cervical myelopathy with or without peripheral neuropathy is the most common manifestation. Spinal MRI of N 2 O-induced myelopathy shows the classic “inverted V” or “rabbit ears” sign. It is characterized by lesions involving dorsal and lateral columns from the cervical to thoracic spine. The cervical spinal cord is the most frequently involved region of the entire spinal cord [5,8-10]. The lesion usually appears as a high-intensity signal on T2-weighted images. However, it appears as a normal signal on T1-weighted images [6,12]. Differential diagnoses include extrinsic compression, inflammatory, infectious, and vascular causes, copper deficiency myelopathy, vacuolar myelopathy in conjunction with chronic HIV infection, sequelae of intrathecal chemotherapy, and tabs dorsalis [6,11,12].
The first step in management is educating patients to stop abusing N2O. Vitamin B12 replacement can be performed using high-dose intramuscular hydroxocobalamin [7,11]. Organizing and understanding detailed clinical patterns of patients will aid in the early diagnosis of N2O-induced myelopathy. We strongly recommend to ask young patients with suspected myelopathy about their history of N2O abuse, especially for those presenting with sensory symptoms and upper cervical long-segment cord lesions. Clinicians should consider the possibility of N 2 O-induced myelopathy in long-segment cervical myelopathy so that treatment can be initiated as early as possible.
Fig. 2.
Cervical spine magnetic resonance image of a 27-year-old man. (A) Sagittal T2-weighted image revealing high signal intensities from C3 to C5 level (arrowhead). (B) Axial T2-weighted image showing high signal intensity in dorsal column (inverted V sign, arrow).
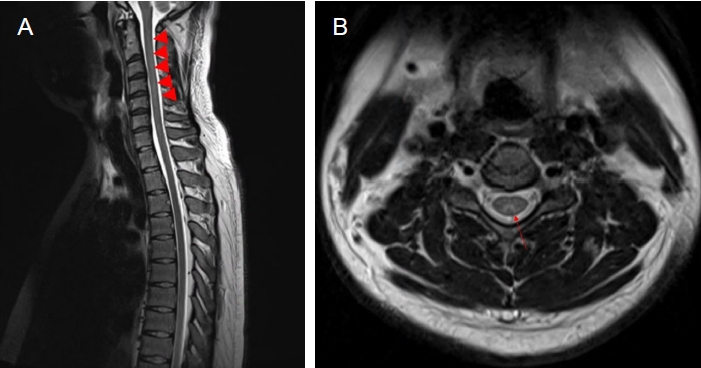
Table 1.
Baseline characteristics of subjects
Table 2.
Laboratory, electrophysiological and imaging findings
REFERENCES
1. Alt RS, Morrissey RP, Gang MA, Hoffman RS, Schaumburg HH. Severe myeloneuropathy from acute high-dose nitrous oxide (N2O) abuse. J Emerg Med 2011;41:378-380.


2. Chiang TT, Hung CT, Wang WM, Lee JT, Yang FC. Recreational nitrous oxide abuse-induced vitamin B12 deficiency in a patient presenting with hyperpigmentation of the skin. Case Rep Dermatol 2013;5:186-191.



3. de Medeiros FC, de Albuquerque LA, de Souza RB, Gomes Neto AP, Christo PP. Vitamin B12 extensive thoracic myelopathy: clinical, radiological and prognostic aspects. Two cases report and literature review. Neurol Sci 2013;34:1857-1860.


4. Patel KK, Mejia Munne JC, Gunness VRN, Hersey D, Alshafai N, Sciubba D, et al. Subacute combined degeneration of the spinal cord following nitrous oxide anesthesia: A systematic review of cases. Clin Neurol Neurosurg 2018;173:163-168.


5. Lan SY, Kuo CY, Chou CC, Kong SS, Hung PC, Tsai HY, et al. Recreational nitrous oxide abuse related subacute combined degeneration of the spinal cord in adolescents - A case series and literature review. Brain Dev 2019;41:428-435.


6. Langan RC, Goodbred AJ. Vitamin B12 deficiency: Recognition and management. Am Fam Physician 2017;96:384-389.

7. Dong X, Ba F, Wang R, Zheng D. Imaging appearance of myelopathy secondary to nitrous oxide abuse: a case report and review of the literature. Int J Neurosci 2019;129:225-229.


8. Jiang J, Shang X. Clinical-radiological dissociation in a patient with nitrous oxide-induced subacute combined degeneration: a case report. BMC Neurol 2020;20:99.



9. Swart G, Blair C, Lu Z, Yogendran S, Offord J, Sutherland E, et al. Nitrous oxide-induced myeloneuropathy. Eur J Neurol 2021;28:3938-3944.


10. Savage S, Ma D. The neurotoxicity of nitrous oxide: the facts and "putative" mechanisms. Brain Sci 2014;4:73-90.